An Addition Reaction Can Best Be Described as
Addition reactions are typical of unsaturated organic compounds ie alkenes which contain a carbon-to-carbon double bond and alkynes which have a carbon-to-carbon triple bondand aldehydes and ketones which have a carbon-to-oxygen double bond. Curtius Rearrangement or Curtius Reaction.
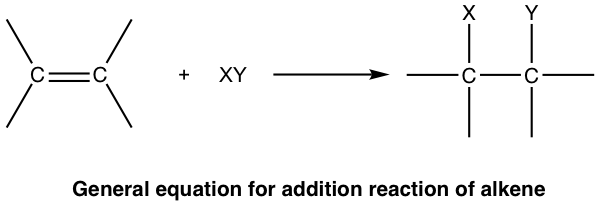
10 2 Reactions Of Alkenes Addition Of Hydrogen Halide To Alkenes Organic Chemistry I
Tap again to see term.

. Addition due to excess HBr present. A a hydrogen reacts with oxygen to produce CO2 H2O and energy. Da single reactant splits into two products.
Addition of a HX to an Internal Alkyne. Polar protic solvents may be used to hinder nucleophiles thus disfavoring E2 S n 2 from occurring. C 2 H 6 C l 2 H C l C 2 H 5 C l is best described as.
It occurs when atoms are added to a compound containing a double or triple bond. RCON3 R-NCO N2. This product will contain all the atoms that were present in the reactants.
Addition due to excess HX present. There are two broad classes of addition reactions. Addition of hydrogen halide to an alkyne.
Initiation propagation and termination. B a single reactant undergoes reorganization of its chemical bonds producing an isomer of the reactant. To make wine grape juice is fermented to produce alcohol.
1 2858 kJ 3 5716 kJ. C increases the reaction rate and has a higher activation energy D increases the reaction rate and has a lower activation energy 11For a given chemical reaction the addition of a catalyst provides a different reaction pathway that A It is endothermic and entropy decreases. The addition reaction is the combination of two or more atoms or molecules in order to form a large molecule.
No other or by- products are formed in an addition reaction. Addition polymerization also known as free-radical polymerization has three stages. Curtius reaction involves the heating of an acyl azide which loses nitrogen and then rearranges to an isocyanate.
The reaction can be initiated by the thermal decomposition of an unstable initiator molecule such as a peroxide to produce free radicals eg benzoyl peroxide decomposes via a two-stage process that yields phenyl radicals and CO 2. Btwo reactants combine to form one new product with no extra atoms. Well take a look at a mechanism involving solvolysis during an E1 reaction of Propanol in Sulfuric Acid.
HI HBr HCl HF. An Introduction to Organic Synthesis. Addition of Hydrogen Halide to an Alkyne.
Chapter 6 Exhibit 6-9 Use the reaction energy diagram below to answer the following questions. The medium can effect the pathway of the reaction as well. B It is endothermic and entropy increases.
Addition reaction any of a class of chemical reactions in which an atom or group of atoms is added to a molecule. Electrophilic and nucleophilic depending on the nature of the first incoming species. Protonation occurs on the more stable carbocation.
Addition of HX to Terminal Alkyne. The reaction often can be stopped after addition of 1 equivalent of HX but reaction with an excess of HX leads to a dihalide product. Tap card to see definition.
This reaction is shown below. An addition reaction can best be described as a reaction in which Question 38 options. Atoms can also be added to molecules in an addition reaction.
A substitution reaction can best be described as a reaction in which. Acid catalyzed dehydration of secondary tertiary alcohols. For example Hydrohalogenation halogenation or polymerization of an alkene.
Tap card to see definition. Click again to see term. Click card to see definition.
We will study three main types of reactions - addition elimination and substitution. Most addition reactions are limited to molecules with unsaturation that have either double bonds or triple bonds. An addition reaction in organic chemistry is in its simplest terms an organic reaction where two or more molecules combine to form a larger one the adduct.
If the reaction is performed in an alcoholic or aqueous medium the isocyanate further reacts to form urethane amine or substituted urea. A rearrangement reaction can be best described as a reaction in which a Two reactants combine to form one new product with no extra atoms b A hydrocarbon reacts with oxygen to produce carbon dioxide water and energy c Two reactants exchange atoms to give two new products. Addition reactions occur when an atom or a group of atoms is combined with another species to generate a larger molecule the adduct.
Fermentation can refer to the conversion of sugar to alcohol using yeast a fungus. Addition reactions occur with unsaturated compounds. A substitution reaction B.
2 H2g O2g 2 H2O 5716 kJ What is the approximate H for the formation of 1 mole of H2O. Yields a geminal dihaloalkane. The process of fermentation produces items such as wine beer and yogurt.
John McMurry Organic Chemistry 8th ed. This large molecule is known as an adduct. For example reaction of 1-hexyne with 2 equivalents of HBr yields 22-dibromohexane.
It is a reaction in which one molecule combines with another to form a larger molecule with no other products. C a single reactant splits into two products. The reaction depicted in this reaction energy diagram can best be described as.
Refer to Exhibit 6-9. An addition reaction occurs when two or more reactants combine to form a single product. Up to 24 cash back 36An addition reaction can best be described as a reaction in which Aa single reactant undergoes reorganization of its chemical bonds producing an isomer of the reactant.
Click card to see definition. Unsaturated hydrocarbons undergo addition reactions. In organic chemistry an addition reaction is an organic reaction in which two or more molecules combine to generate a bigger one the adduct.
Molecules with carbonhetero double bonds such as carbonyl CO or imine CN groups can be added because they have double-bond character as well. In the field of chemistry an addition reaction can be described as a type of reaction in which a molecule combines with another molecule to form a large molecule. C6H12O6 C 6 H 12 O 6 2CO2 2C2H5OH 2 CO 2 2 C 2 H 5 OH energy.
A slow exothermic reaction a fast exothermic reaction a slow endothermic reaction a fast endothermic reaction ANS. An addition reaction. Reactivity order of hydrogen halides.
Ca hydrogen reacts with oxygen to produce CO2 H2O and energy. All of the following are example of addition reactions of alkenes except. D A single reactant undergoes reorganization of its chemical.
With reference to energy the reaction A B AB can best be described as 1 endothermic having a ΔH 2 endothermic having a ΔH 3 exothermic having a ΔH 4 exothermic having a ΔH 28. Hydrogen adds to the carbon with the greatest number of hydrogens the halogen adds to the carbon with fewer hydrogens. These addition reactions can be classified as follows.
A chemical transformation involving the breaking and forming of bonds occurs during an addition reaction.

Wide Squat Strengthening Yoga Yoga Sequences Yoga Poses

Orange Extract Beer Cookies Beer Brewing Home Brewing
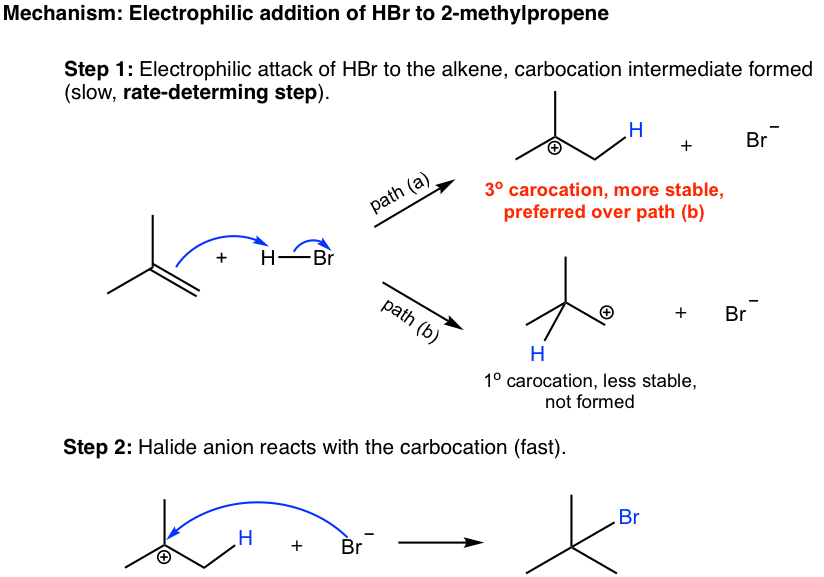
10 2 Reactions Of Alkenes Addition Of Hydrogen Halide To Alkenes Organic Chemistry I

Because Moms Can T Live On Coffee Alone Wine Glass Baby Etsy Birthday Girl Wine Glass Wedding Wine Glasses Wine Glasses Gift

Atomic Theory And Atoms Notes Studyblr Chemistry Studygram Notes Study Notes School Study Tips Life Hacks For School

Lattice Energy I Ionic Compounds A Basic I Introduction I Lattice Energy Of Ionic Compounds A Basic Youtube Ionic Compound Ionic Bonding Ionic
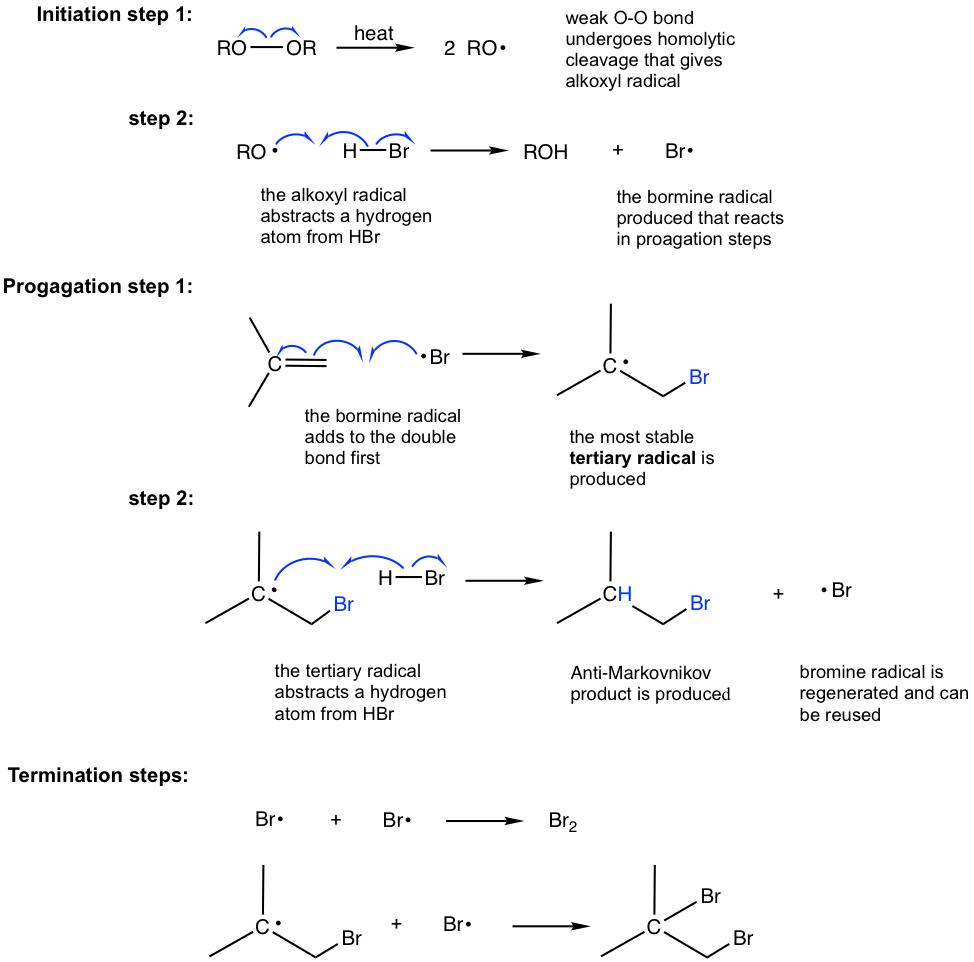
10 2 Reactions Of Alkenes Addition Of Hydrogen Halide To Alkenes Organic Chemistry I

The Emotions Toolbox Emotional Self Regulation In Physical Education Self Regulation School Psychology Tools Emotional Regulation

Pin On Alkene Reactions With Practice Problems

Dnd 5e Homebrew Dungeons And Dragons Game Dnd 5e Homebrew Dungeons And Dragons Homebrew

Pin On Dude I Want That December 2013

Alarm Points Laminated Chart Sku Alarm Energy Medicine Acupressure Points Chart

Pin By Heidi Wagner On Creative Ideas In 2021 Creative People Creative Ken Robinson

Desert Landforms 1 Types Of Deserts Wind School Landforms Geography Revision Geography For Kids Physical Geography

Finding Number Of Oximes Formed Numbers Form Learning

Pin On Alkene Reactions With Practice Problems

Why Aldehydes Are More Reactive Than Ketones Nucleophilic Addition Reaction Ketones Electron Donating Groups Ap Chemistry

Difference Between Oxidation And Reduction Redox Reaction Chemistry Basics Chemistry Lessons Chemistry Education

Comments
Post a Comment